Habib™
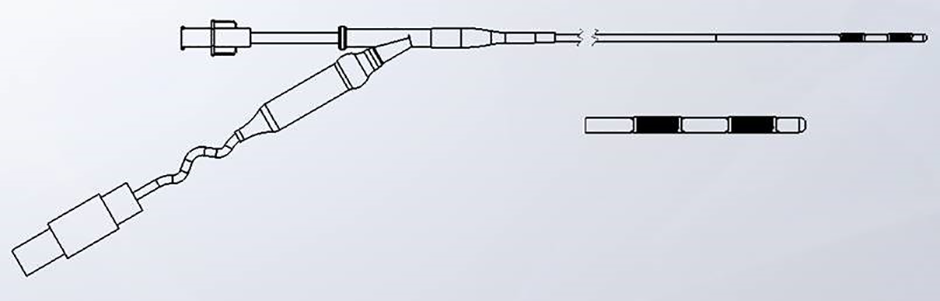
EndoHPB Bipolar Radiofrequency Catheter
The Habib™ EndoHPB Catheter is the first Radio Frequency (RF) ablation catheter indicated in the U.S. for malignant or benign tissue ablation in the pancreatic and biliary tract.
Explore
When surgery is not an option for patients with difficult-to-treat cancers, the Habib™ EndoHPB Catheter enables you to perform:
- Partial or complete ablation of malignant or benign tissue in the pancreatic and biliary tracts.
- Endoscopic biliary drainage or decompression prior to stent placement or afterwards to clear an occluded stent.
Endoscopic biliary RF ablation with the Habib™ EndoHPB Catheter prior to stent placement has been shown to prolong metallic biliary stent patency by an average of 13% (from 8.4 months to 9.5 months)1
Product Details
| Device Specification | Intended Benefits |
|---|---|
| 180cm useable length, 8Fr (2.7mm) diameter | Enables biliary access through a 3.2mm working channel duodenoscope |
| 2, 8mm stainless steel ring electrodes | Produce ablation depths 3-4mm from the wall of the catheter. Resulting ablation zone is 25mm ± 3mm long by 9mm ± 2mm wide (1) |
| Compatible with commonly available RF generators and endoscopes with a working channel of 3.2mm or greater | Does not require the purchase of dedicated capital equipment |
| Bipolar RF Device | Use of adapter cable enable bipolar RF ablation and avoids the need for electrode grounding pads |
Features and Benefits
- Treatment alternative that may prolong patency and restore biliary drainage1
- The Habib EndoHPB Catheter may provide an option to restore biliary drainage in patients who may outlive the patency of their metallic biliary stents.2
- Clinical data from one study suggests that RFA with the Habib EndoHPB Catheter for occluded SEMS may improve mean stent patency time compared to plastic stent insertion.2
Minimal Capital Investment
The Habib™ EndoHPB Catheter is compatible with many commonly available electrosurgical generators and endoscopes with a working channel of 3.2mm or greater, eliminating the unique capital expenditure. There is also no need for electrode grounding pads when using our adaptor cable.
| Generator | Adaptor Cable Bipolar | Recommended Generator Settings |
|---|---|---|
| ERBE ICC200, ICC300, ICC350 | 5420 or 5700 | Bipolar Mode Coagulation mode 7-10 Watts 90 seconds |
| ERBE VIO* 200 or 300 (D/S) | 5420 or 5700 | Soft Coag Mode Effect 8 7-10 Watts 90 Seconds |
| ERBE VIO* 3 | 5100 or *5500 or 5700 | Soft Coag Bipolar Mode Effect 1.4 - 2.0 (Low-High) 90 Seconds |
| Gi4000 | 5420 | Bipolar Mode 22-32 Watts 120 Seconds |
| KLS Martin MaXium* | 5100 | Micro Coag Mode 9-12 Watts 90 Seconds |
| Olympus ESG-100 | 5100 | Soft Coag Mode Level 22-32 120 Seconds |
Ease of Use
- No need for dedicated capital equipment
- Compatible with most electrosurgical generators
- Simple time and power algorithm delivers consistent results3
- One probe size, each ablation produces an ablation zone approximately 25mm x 3.5mm deep (at 10W, 90s).3
Ordering Information
| Product Number | Product Description | Specifications |
|---|---|---|
| M00500070 | Habib EndoHPB Bipolar RF Catheter (180cm, 8F) | Single-Use |
| 5420 | Bipolar Adapter Cable | Reusable |
Contact Information and Ordering
For product orders or complaints, please contact: 1-888-272-1001
Reimbursement
Physician & Patient Resources
REFERENCES
1. Liang, H. et al, “Metal Stenting with or without Endobiliary Radiofrequency Ablation for Unresectable Extrahepatic Cholangiocarcinoma”, Journal of Cancer Therapy, October 2017.
2. Kadayifci A. et al., Endoscopy 2016;48:1096-1101
3. DN VAL-R-020










